The Bottom Line
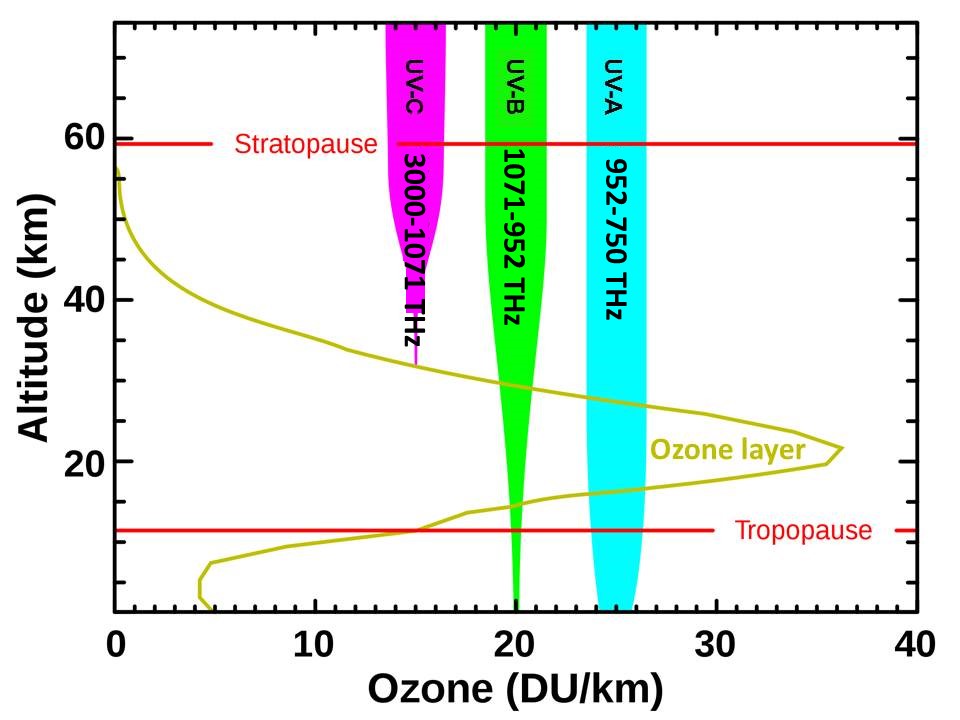
The atmosphere absorbs the highest energy ultraviolet solar radiation before it can damage life on Earth. How much of this high-energy radiation reaches each altitude determines the physical, chemical, and thermal structure of Earth’s atmosphere. The ozone “layer” is not a static layer of gas, but a region in the atmosphere where sufficient high-energy solar ultraviolet radiation is available to form and destroy ozone in an endless cycle (The Chapman Cycle), each primary step of which warms the stratosphere. The lifetime of a molecule of ozone in the stratosphere can be as short as seconds in the upper stratosphere and is about 22 days in the lower troposphere. When the normal amount of ozone is depleted for whatever reason, some of the solar ultraviolet energy that drives this cycle is not absorbed in the stratosphere but penetrates to Earth where oceans absorb it very efficiently, cooling the stratosphere and warming Earth.
Major explosive volcanic eruptions form aerosols in the lower stratosphere that reflect and disperse sunlight, cooling Earth approximately 0.5oC for a few years. These explosive eruptions also deplete ozone, warming Earth in winter, but cooling caused by the aerosols predominates. Effusive, basaltic volcanic eruptions, even small ones, on the other hand, warm Earth by depleting ozone just as much as large explosive eruptions but not exploding gases high enough to form substantial aerosols. Climate throughout Earth’s history has been controlled primarily by the mix of explosive and effusive volcanism, which is determined by the details of plate tectonics, the movement of large segments of Earth’s surface relative to each other.
Global warming between 1970 and 1998 of approximately 1oC (1.8oF) was caused by human manufactured chlorofluorocarbons that depleted ozone. Through the Montreal Protocol, we have stopped such manufacturing and ozone depletion is decreasing, but Earth will not cool down. We have increased Earth’s thermostat and must live with the results as we approach a new thermal equilibrium.
Greenhouse gases did not cause this global warming and reducing carbon emissions will not affect climate, although it might reduce ocean acidification.
Specific conclusions explained in detail on this website are:
- The atmosphere absorbs most high-energy ultraviolet solar radiation before it can reach Earth. When the normal amount of ozone in the lower stratosphere is depleted, more high-energy ultraviolet radiation than usual reaches Earth where it is absorbed most efficiently by oceans that cover 71% of Earth’s surface.
- An increase in sunburning (erythemal) radiation has been well observed since 1970 and is clearly associated with widespread ozone depletion, greatest in the Antarctic ozone hole.
- Mean air temperature in the lowermost atmosphere appears to be determined primarily by mean temperature of the mixed layer at the ocean’s surface, not visa versa.
- Temperature of matter is a function of the oscillatory energy contained within matter. Global warming is about changes in radiation that are effective at increasing internal energy within matter, not about the amounts of available energy integrated across a broad spectral band.
- According to the Planck postulate, energy in radiation is equal to the frequency of oscillation contained in the radiation times a constant. The oscillatory energy involved in solar ultraviolet radiation reaching Earth with frequencies around 967 terahertz (wavelength=310 nanometers, energy=4 electron volts, eV) is 48 times greater than the oscillatory energy of infrared radiation absorbed by greenhouse gases such as carbon dioxide with a frequency of 20.1 terahertz (wavelength=14.9 micrometers, energy=0.083 electron volts).
- Temperature in a gas is proportional to the mean velocity of all molecules within the gas. When solar ultraviolet energy causes photodissociation, the energy of oscillation contained in the molecular bonds broken is converted into translational kinetic energy of the separating molecular pieces, heating the gas. Infrared radiation absorbed by greenhouse gases increases the internal oscillatory energy of the molecule but has limited direct effect on the temperature of the gas.
- The observed rise in mean surface temperatures in the northern hemisphere by approximately 1oC between 1970 and 1998 (HadCRUT4.2) (Jones et al., 2012) appears to have been caused primarily by depletion of the ozone layer due to chlorofluorocarbons manufactured by humans.
- Atmospheric concentrations of CO2 from 150 to 800,000 years ago may simply be a proxy for ocean heat content, reflecting the solubility of CO2 as a function of ocean temperature.
- The rise in concentrations of carbon dioxide (CO2) during the 20th century may simply result from anthropogenic emissions plus increasing ocean heat content.
- International concern over the Antarctic ozone hole led, in 1989, to the Montreal Protocol on Substances that Deplete the Ozone Layer that was ratified by 196 states plus the European Union, one of the most widely ratified treaties in United Nations history.
- By 1992, emissions of chlorofluorocarbons had been decreased sufficiently to slow increases in temperature caused by ozone depletion. But ozone depletion caused by the 1991 eruption of Pinatubo volcano made this change in trend less clear until 1998.
- Mean global temperatures have remained nearly constant since 1998 as ozone depletion begins to recover. But ocean heat content continues to increase because ozone has remained depleted.
- The effects of chlorofluorocarbons and related chemical processes depleted ozone approximately 3%.
- This depletion is expected to recover slowly, perhaps by 2070, because of the long lifetime of chlorofluorocarbons in the atmosphere.
- Complete recovery of the ozone layer will not cool Earth. Ozone depletion warms the ocean; when ozone is returned to normal levels, the warmer ocean remains.
- Volcanic eruptions of all sizes deplete ozone by as much as 6%, causing warming. Ozone levels generally return to normal within one decade.
- But large explosive volcanic eruptions, such as Pinatubo in 1991, form sulfuric-acid and water aerosols in the lower stratosphere, reflecting and dispersing sunlight, causing net global cooling of as much as 0.5oC for up to 3 years.
- Effusive, basaltic volcanic eruptions, even very small ones, deplete ozone as much as 6% but do not eject sulfur dioxide high enough in the atmosphere to form aerosols, causing net global warming for up to a decade.
- Changes in climate throughout geologic time appear to be controlled primarily by the balance of explosive volcanic eruptions that cool the ocean by forming aerosols in the lower stratosphere and effusive basaltic volcanic eruptions that warm the ocean by depleting ozone. This balance is determined by the distribution of volcanism types and amounts associated with plate tectonics, the motion of large plates around the surface of Earth.
- The record-setting warming and drought in 2012 over much of North America appears to have been caused by ~14% depletion of ozone resulting from anthropogenic emissions plus the eruptions of Eyjafjallajökull (2010) and Grímsvötn (2011) in southern Iceland.
- The Dust Bowl warming and drought during the1930s appears to have been caused by a highly unusual sequence of seven moderate volcanic eruptions between 1931 and 1933 in Indonesia, Japan, Kurile Islands, Kamchatka, Alaska, Guatemala, and Chile.
- Earth’s atmosphere is heated primarily by solar energy sufficient to cause photoionization and photodissociation of gases. Absorption by any gas of energy not sufficient to cause photoionization or photodissociation appears to have little effect on warming the atmosphere.
- Reducing carbon dioxide (CO2) emissions is not likely to reduce global warming although it may reduce ocean acidification.
- Several assumptions made by scientists more than a century ago concerning the flow of energy via radiation and used to quantify greenhouse gas theory have not been tested in the laboratory and do not appear to be physically correct.
- For example, the temperature of matter cannot be increased by absorbing radiation from colder bodies of matter, as assumed by greenhouse-gas theory. You do not stand around a cold stove to get warm.
- It is widely assumed that electromagnetic radiation (light) travels in space in a manner similar to waves traveling in matter. Waves deform matter but there is no matter in space. Radiation in space is simply an electromagnetic field containing oscillatory energy that is not visible until it interacts with matter. This energy field causes the bonds holding molecules of gas together to resonate, absorbing only resonant frequencies from the surrounding field.
- Electromagnetic radiation might be described most precisely as field-particle-wave triality or vacuum-gas-solid triality to emphasize that the physical properties observed for light are distinctly different in space, in atmospheres, and in matter.
- It is well known that the ozone layer protects life on Earth from solar, high-energy, ultraviolet radiation. When the normal amount of ozone is depleted, more ultraviolet energy reaches Earth, cooling the lower stratosphere, warming the upper troposphere, raising the height of the tropopause, and warming Earth primarily by warming the oceans.
Each of these conclusions is explained in detail on this website.