Properties of the Electromagnetic Spectrum
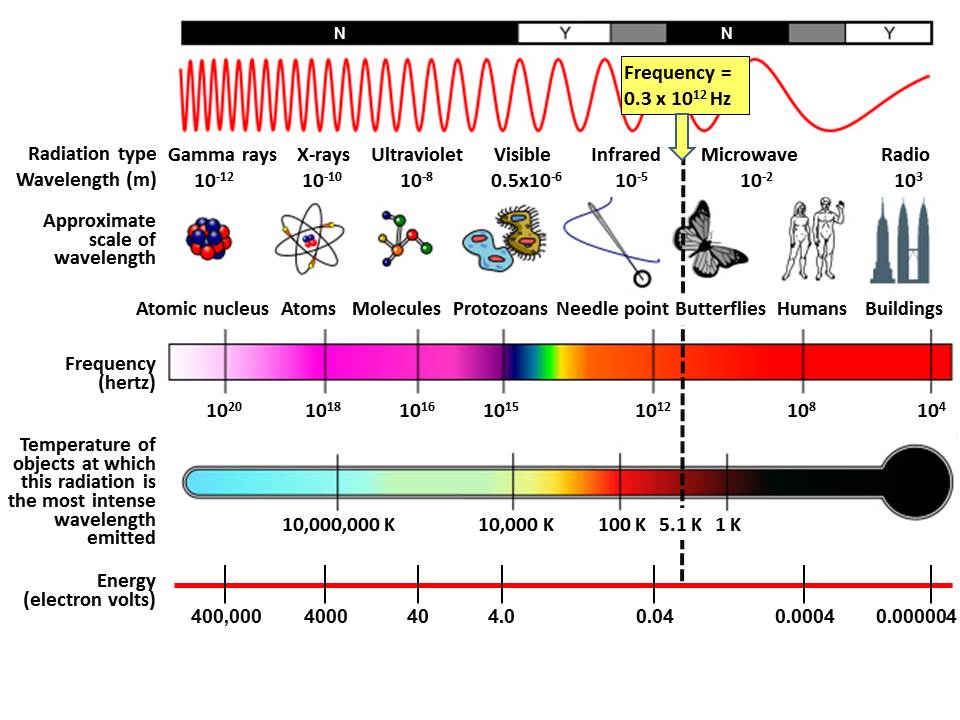
The top bar shows whether the radiation penetrates earth’s atmosphere relatively efficiently (Yes or No). Next the wavelengths in meters, using scientific notation. Notice the spectrum goes from the longer Radio waves, at 103 meters, all the way to the shorter Gamma Rays, at 10-12 meters. Next the various wavelengths are compared to sizes found in everyday life. Then the frequency of oscillation in cycles per second, hertz. Note that visible light has frequencies in the range of 6 x 1014 hertz, 600 terahertz. Next the temperature of objects at which this radiation is the most intense wavelength emitted according to Planck’s law. Finally the energy contained in radiation at this frequency according to the Planck postulate energy equals frequency times the Planck constant.
Based on Wikipedia. Energy scale added.