Frictionless Atomic Oscillator
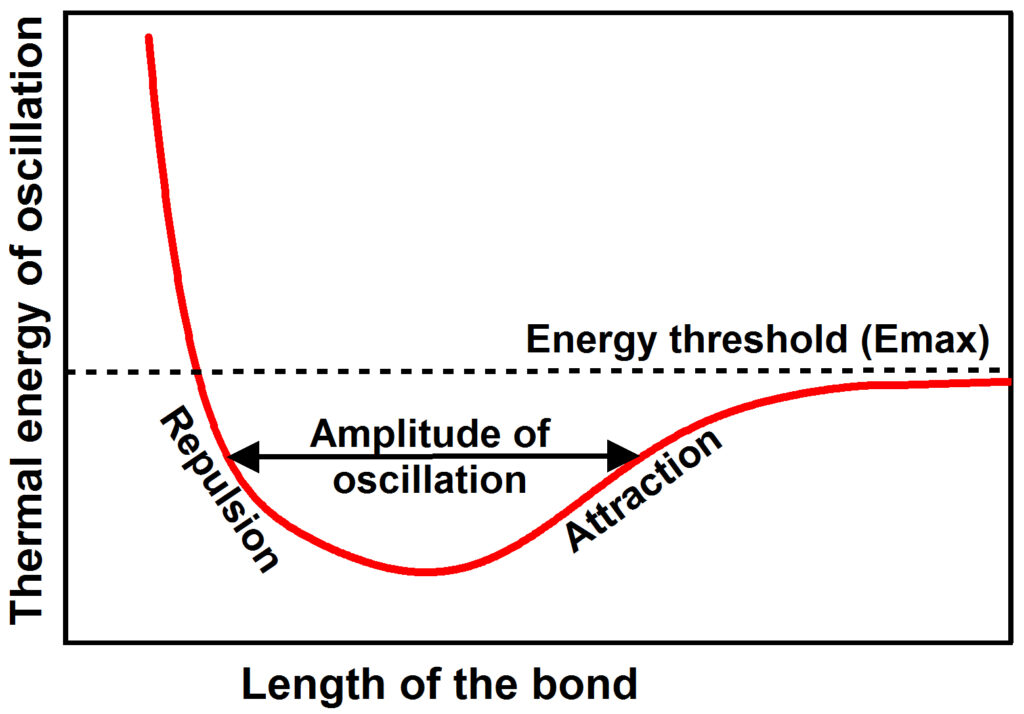
The chemical bonds that hold atoms together to form matter are not rigid. They are observed to oscillate about an energy minimum between electrodynamic repulsive forces pushing the atoms apart and electrodynamic attractive forces pulling the atoms together. As the thermal energy of oscillation increases, the amplitude of oscillation increases until at some energy threshold (Emax) the bond comes apart. Energy (frequency) may increase in discrete steps as higher and higher normal modes (frequencies) of oscillation are activated. One such anharmonic atomic oscillator exists for every normal mode of oscillation of every degree of freedom of every bond in matter. Each oscillator has a characteristic resonant frequency and an amplitude of oscillation, the latter of which increases with increasing temperature (thermal energy). Because the oscillator is asymmetric, the average length of the bond increases with increasing thermal energy so that the volume of the matter expands with increasing temperature, something that is well observed.