Absorption by Gases in the Atmosphere
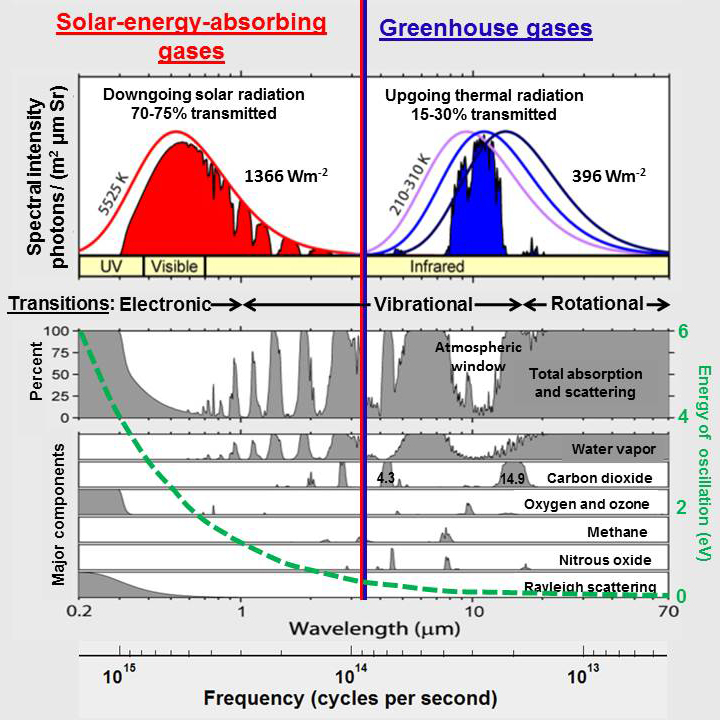
Greenhouse gases such as carbon dioxide and methane absorb only a very small amount of the broad spectrum of infrared heat (energy) radiated by Earth. Based on a figure by Rohde (2007).
The red line shows the spectral radiance, the amount of radiation at each wavelength, for solar radiation at the top of Earth’s atmosphere according to Planck’s law assuming the Sun has a surface temperature of 5525K. The purple, blue, and navy lines show the spectral radiance for infrared energy radiated by Earth at a temperature of 210K, 260K, and 310 K respectively The solar and terrestrial curves have been normalized to have the same height. Peak spectral radiance of solar radiation at the top of the atmosphere is actually 69 times larger than the peak spectral radiance for terrestrial radiation. The blue shaded area shows the spectral radiance for terrestrial radiation reaching space while the red shaded area shows the spectral radiance for solar radiation reaching Earth.
The gray shaded areas show the percentage of absorption and Rayleigh scattering that occur at each wavelength. Note that water vapor absorbs energy over a wide variety of wavelengths while carbon dioxide only absorbs energy in a few narrow wave bands. The band around 4.3 micrometers (μm) does not overlap with a water vapor absorption band but contains little infrared energy. The band around 14.9 μm overlaps absorption by water vapor substantially, although the spectral lines (Rothman et al., 2013) for the two gases do not overlap much in detail. Concentrations of water vapor range from 0.1 to 5% while concentrations of CO2 are less than 0.04%.
Note that absorption by photodissociation of ozone is along a continuum at wavelengths below 0.35 μm and that much of what we think of as Rayleigh scattering my be related to photodissociation.
The green dashed line shows the Planck postulate (E=hν) emphasizing that the energy contained in radiation increases with decreasing wavelength, increasing frequency.